States of Matter: Hands-On Activities about Liquids
In this part of our States of Matter unit we talked about some of the properties of liquids.
A few days ago, I shared some of activities as we discussed gases. You’ll find that here: States of Matter-Hands-On Activities about Gases. We continued to talk about the structure of matter. Liquids have a definite volume, but no definite shape.
Not only did we talk about some of the characteristics of liquids:
- they take up space,
- have weight,
- flow,
- change to fit the container and so forth,
but we also went on to explore some of the properties of water such as adhesion, cohesion, surface tension, and things like that. Once again, the kids loved all of the hands-on experiments and activities! Let me share a few of those with you now!
We had some good discussions about ooblek (cornstarch and water) and debated whether it is a solid or a liquid… Sometimes it seems solid (like when you press your thumb down on it or squeeze it in your fist. Other times it flows like a liquid.
We talked about polymers, those long chains of molecules that cause ooblek to act as it does. I used the analogy of ooblek having molecules like spaghetti while other liquids have molecules that are more like peas.
Cornstarch: Is it a solid or a liquid?!!

Is shaving cream a solid or a liquid?
We went on to study some of the properties of liquids. (These are in our States of Matter packet.)

We took several glass graduated cylinders and filled them with water. The narrowest cylinder showed the most adhesion. We also talked about the meniscus.
Demonstrating Cohesion:
How close can you drag to drops of water before they “jump” together? Why do they “jump” together?
For this activity we used the printout from MiddleSchoolChemistry.com, covered with wax paper.
To demonstrate cohesion, we also printed out the Race Drop Raceway from MiddleSchoolChemistry.com. We placed the paper on cardboard and then covered it with a sheet of wax paper. We taped wax paper to the back of the cardboard to it wouldn’t move.
Watch as the droplet drags behind and then catches up.
This little video demonstrates cohesion better than a still photo!!
Demonstrating the effect of gravity on water; water pressure increases with depth:
We used a plastics bottle and colored water. We created holes in the plastic bottle by using a hammer and a nail.
Molecule movement is affected by temperature:
We had two small jars. One had ice cold water, the other had boiling hot water. We added one drop of each color dye into the jars and observed. (The cold water was slow to mix. In the hot water the colors mixed together rapidly.)
Graphing Activity, The World’s Waters:
We also did another graphing activity about the world’s waters. (We did a graphing activity about the gases in air as well in this unit.)
From here we went on to talk about the Changes in States of Matter. I’ll share those activities in the next post!! Here is the link to our 50+ page States of Matter Packet. 🙂
After finishing this unit, we went straight on to our next unit, the Physical and Chemical Properties of Matter.
In the Properties of Matter Unit we are studied:
- Matter: Elements, Compounds, Mixtures
- Organization of the Periodic Table
- Molecular vs. Structural Formulas
- Describing Matter: Physical and Chemical Properties
- Density Activities – Mass÷Volume
- Mixtures: Solutions, Colloids and Suspensions
Chemistry BUNDLE purchase these 6 units together
- Introduction to Chemistry – Size of Atoms Mini-Unit
- States of Matter Packet
- Properties of Matter Packet
- Electricity & Circuits Packet
- Chemistry Packet
- pH Scale – Acids and Bases Worksheet Packet
Chemistry BUNDLE of 6
$27.99


$2.99 Introduction to Chemistry – Size of Atoms Mini-Unit
$5.99 States of Matter Packet (50+pages)
$7.99 Properties of Matter Packet (now close to 100 pages)
$6.99 Electricity & Circuits Packet (30+pages)
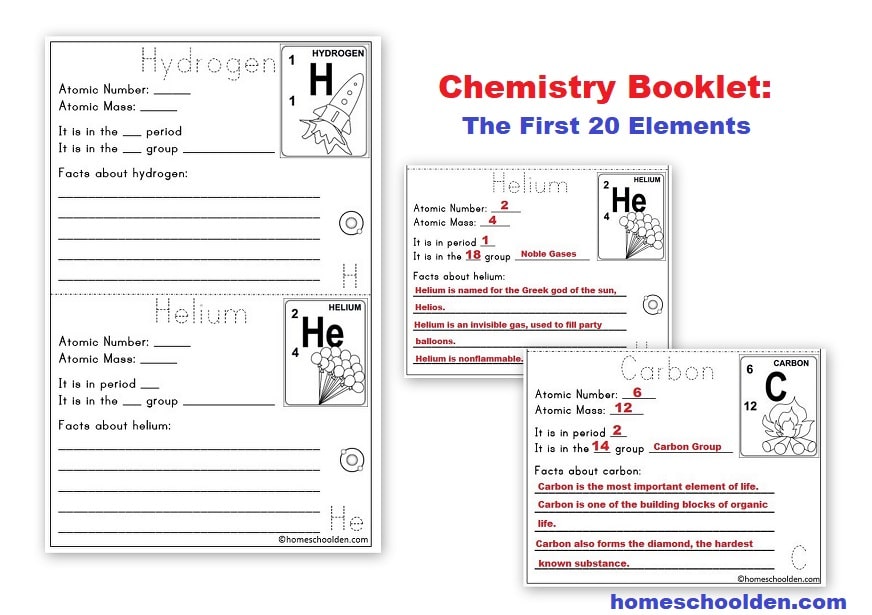
$8.99 Chemistry Packet – An introduction to the Periodic Table (150 pages) – Learn how the periodic table is organized and find out more about the different groups of the periodic table (Alkali metals, alkaline earth metals, halogens). Learn what valence electrons are and do some fun activities to create Lewis structures. Learn about the first twenty elements as well as elements with unusual names. Plus, learn about electron configuration and more! (We did this primarily in middle school, but did electron configuration in high school again.)
$2.99 pH Scale – Acids and Bases Worksheet Packet (25 pages) – Includes material for younger students (elementary/middle school) and older (high school) students.
$7.99 Ions and Isotopes Chemistry Unit (125 pages) This unit is an introduction to ions (which have fewer or more electrons) and isotopes (which have fewer or more isotopes). This unit should be done after students have completed the Chemistry Unit. (middle school/high school)
$9.99 Organic Chemistry Unit (100+ pages) Organic chemistry is the chemistry of carbon-containing molecules. In this unit, we study many of the organic compounds that include carbon and hydrogen (hydrocarbons), carbon-carbon molecules and more! This unit covers hydrocarbons, functional side groups and the four classes of macromolecules: carbohydrates, proteins, fats and nucleic acids. *high school level unit
Click on the link to see more details about each of these units:
Introduction to Chemistry – Size of Atoms Mini-Unit
Chemistry Packet: The Periodic Table
pH Scale – Acids and Bases Worksheet Packet
See you again soon here or over at our Homeschool Den Facebook Page. Also, don’t forget to subscribe to newsletter.
Disclosure: Please note that some of the links in this post are affiliate links, and at no additional cost to you, I will earn a commission if you decide to make a purchase.
Other chemistry posts that may be of interest:
Chemistry Packet: Introduction to the Periodic Table
I am excited to share our new chemistry unit with you! As you know, we love hands-on activities and I want to show you some of the fun ways we explored the periodic table and touched on topics like valence electrons, Bohr Diagrams, Lewis Diagrams (electron dot diagrams), ions, isotopes, and more!
Some of the topics we explored included:
- Building the Periodic Table
- Bohr Diagrams & Understanding Valence Electrons
- Periods, Groups & Families
- hydrogen & the alkali metals, alkaline Earth metals, halogens, noble gases
- Atomic Number, Atomic Mass & Chemical Symbols
- Lewis Diagrams
- Metals, Metalloids and Nonmetals
- Unusual Element Symbols
- Trends of the Periodic Table
- Electron Configuration
We did this unit together when the kids were 10, 12 and 14. I think this unit is best for middle school and up (perhaps as a supplement to for high schoolers being introduced to chemistry for the first time).
Click here to see more details about our Chemistry Packet:
- Chemistry Experiments for Kids (Grade 2) – Matter is Neither Created Nor Destroyed — Acids and Bases
Don’t miss our FREE chemistry packet from last year:
- Chemistry Experiments for Kids (Grade 2) – Mixtures, Chromatography, DNA Kit
- Chemistry Unit: Periodic Table – The chemistry unit we did last year as we learned about the main groups of the periodic table
- Alkali Metals — reactive
- Alkaline Earth Metals
- Halogens
- Noble Gases
- Chemistry Unit: Bohr Diagrams (this post has a free printable)
- Explosion of Colors in Milk Experiment and Other Chemistry Fun!
- Chemistry Unit: The Size of Atoms
- States of Matter: Solid, Liquid, Gas — Learning Activities
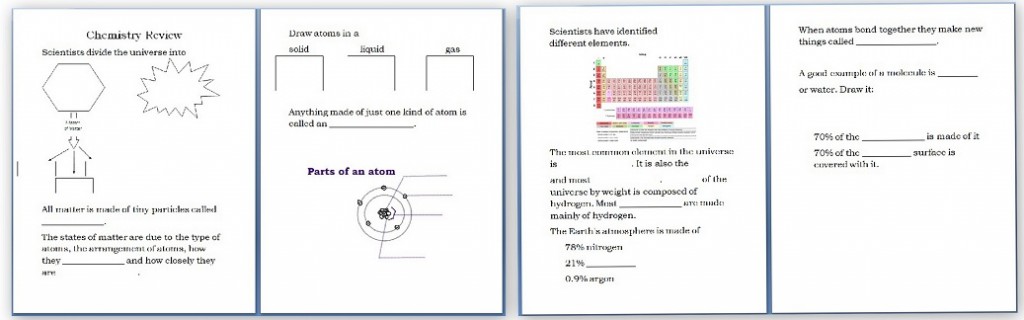
- Chemistry: Molecule Movement Experiment and Chemistry Review Worksheet These notebook pages are free.
- Science Experiments: Water Molecule Attraction
- Building Molecules Chemistry Activity This also has some free notebook pages about building molecules:

See you again soon here or Homeschool Den Facebook page. Don’t forget to Subscribe to our Homeschool Den Newsletter! ~Liesl