Changes in States of Matter: Sublimation and Deposition Activities with Dry Ice!
Sublimation and Deposition Activities – Making Chemistry Fun!
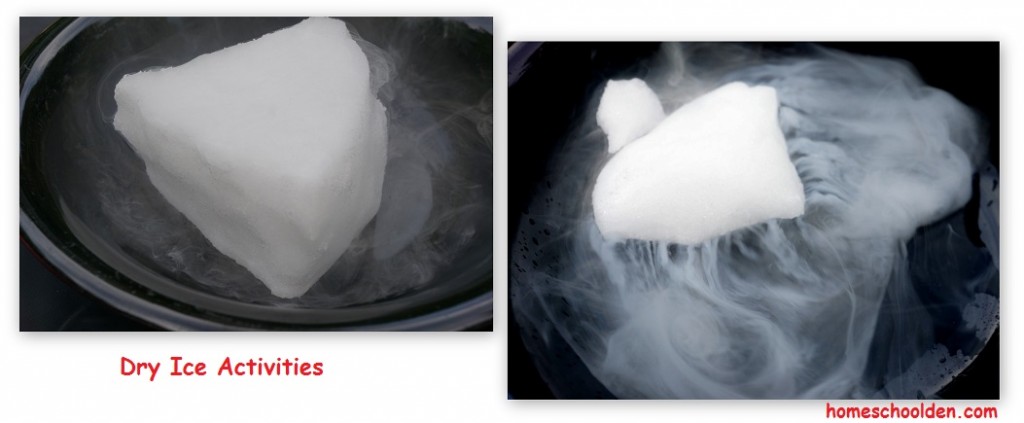
This fall we’ve been studying the states of matter and changes of matter. Today I’m going to share some of our activities as we talked about the changes in states of matter from solid to gas–sublimation and from gas to solid–deposition. It was a day of Dry Ice FUN!! 🙂
While most kids know freezing, melting and evaporation sublimation and deposition are a bit more challenging, that is – until you bring in a huge chunk of dry ice!!
Warning: Always wear gloves when handling dry ice! it is MINUS 109 deg F. That’s cold enough to give you frost bite!
You’ll need a hammer to break the ice into smaller chunks.
I purchased a block of dry ice from the grocery story. It’s held in a separate freezer, so just ask at your local supermarket. Most chains carry it. I brought a small ice chest to put it in.
I set up our work area outside. These activities worked really well because it was a warm humid day.
I wanted to start off with excitement… and had the kids working quietly in the homeschool room. I then tossed a chunk of dry ice in the toilet. I holler out, “Everyone, come quick!! What’s going on…” Then when they were all peering into the toilet bowl, I said… “Whoa… I think it’s sublimation!! Sublimation everyone! Sublimation.” My son was quick to explain to the others “It’s dry ice!!”
So… my picture didn’t turn out very well, but you get the idea!!
Then we went outside where I had everything set up.

Next, the kids poured water into the bowl with the dry ice. That definitely led to a lot of “Wows” and “Ohhs and Ahhs!”

They noticed that the coins then had frost accumulate on top; this is deposition. Solids form on the coin as the humid air is cooled. The coin was soon covered in frost!


Our last activity was to add some dry ice to apple juice. The kids wanted to know why we were doing that. They wondered it was just to watch the apple juice bubbling. I had them wait (and wait) and teased them by saying, “okay, now watch” as the last bit of dry ice melted. “What?! That’s IT?!” they asked, outraged. “Yup,” I said and took a sip. When their dry ice finally melted, they were caught completely by surprise that it was carbonated!!
The next day, we talked more about the changes in states of matter and did a couple of activities for our notebook pages. These are included in our States of Matter Packet. 🙂

After finishing this unit, we went straight on to our next unit, the Physical and Chemical Properties of Matter.
In the Properties of Matter Unit we are studied:
- Matter: Elements, Compounds, Mixtures
- Organization of the Periodic Table
- Molecular vs. Structural Formulas
- Describing Matter: Physical and Chemical Properties
- Density Activities – Mass÷Volume
- Mixtures: Solutions, Colloids and Suspensions
Chemistry BUNDLE purchase these 6 units together
- Introduction to Chemistry – Size of Atoms Mini-Unit
- States of Matter Packet
- Properties of Matter Packet
- Electricity & Circuits Packet
- Chemistry Packet
- pH Scale – Acids and Bases Worksheet Packet
Chemistry BUNDLE of 6
$27.99


$2.99 Introduction to Chemistry – Size of Atoms Mini-Unit
$5.99 States of Matter Packet (50+pages)
$7.99 Properties of Matter Packet (now close to 100 pages)
$6.99 Electricity & Circuits Packet (30+pages)
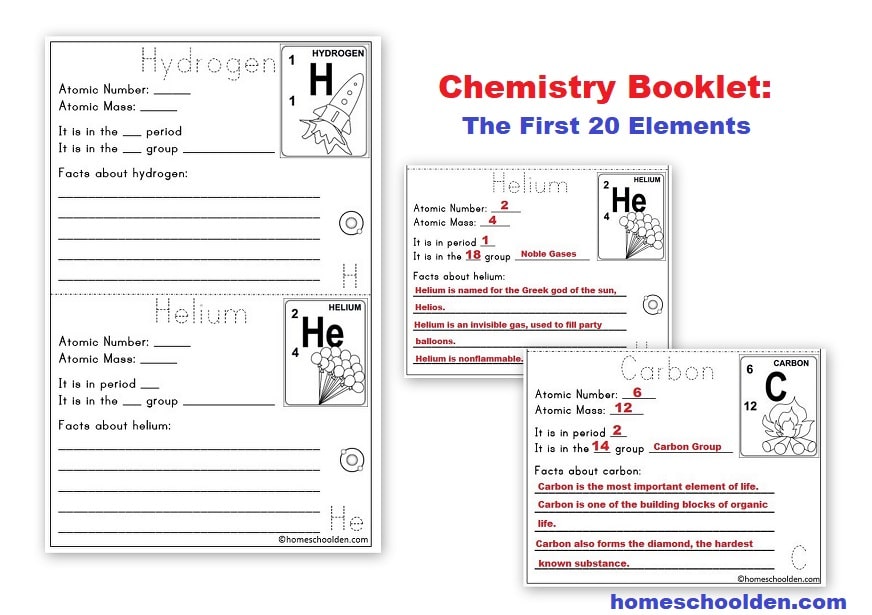
$8.99 Chemistry Packet – An introduction to the Periodic Table (150 pages) – Learn how the periodic table is organized and find out more about the different groups of the periodic table (Alkali metals, alkaline earth metals, halogens). Learn what valence electrons are and do some fun activities to create Lewis structures. Learn about the first twenty elements as well as elements with unusual names. Plus, learn about electron configuration and more! (We did this primarily in middle school, but did electron configuration in high school again.)
$2.99 pH Scale – Acids and Bases Worksheet Packet (25 pages) – Includes material for younger students (elementary/middle school) and older (high school) students.
$7.99 Ions and Isotopes Chemistry Unit (125 pages) This unit is an introduction to ions (which have fewer or more electrons) and isotopes (which have fewer or more isotopes). This unit should be done after students have completed the Chemistry Unit. (middle school/high school)
$9.99 Organic Chemistry Unit (100+ pages) Organic chemistry is the chemistry of carbon-containing molecules. In this unit, we study many of the organic compounds that include carbon and hydrogen (hydrocarbons), carbon-carbon molecules and more! This unit covers hydrocarbons, functional side groups and the four classes of macromolecules: carbohydrates, proteins, fats and nucleic acids. *high school level unit
Click on the link to see more details about each of these units:
Introduction to Chemistry – Size of Atoms Mini-Unit
Chemistry Packet: The Periodic Table

pH Scale – Acids and Bases Worksheet Packet

- States of Matter: Hands-On Activities about Gases
- States of Matter: Hands-On Activities about Liquids
See you again soon here or over at our Homeschool Den Facebook Page. Also, don’t forget to subscribe to newsletter.
Disclosure: Please note that some of the links in this post are affiliate links, and at no additional cost to you, I will earn a commission if you decide to make a purchase.
Other chemistry posts that may be of interest:
Chemistry Packet: Introduction to the Periodic Table
I am excited to share our new chemistry unit with you! As you know, we love hands-on activities and I want to show you some of the fun ways we explored the periodic table and touched on topics like valence electrons, Bohr Diagrams, Lewis Diagrams (electron dot diagrams), ions, isotopes, and more!
Some of the topics we explored included:
- Building the Periodic Table
- Bohr Diagrams & Understanding Valence Electrons
- Periods, Groups & Families
- hydrogen & the alkali metals, alkaline Earth metals, halogens, noble gases
- Atomic Number, Atomic Mass & Chemical Symbols
- Lewis Diagrams
- Metals, Metalloids and Nonmetals
- Unusual Element Symbols
- Trends of the Periodic Table
- Electron Configuration
We did this unit together when the kids were 10, 12 and 14. I think this unit is best for middle school and up (perhaps as a supplement to for high schoolers being introduced to chemistry for the first time).
Click here to see more details about our Chemistry Packet:
- Chemistry Experiments for Kids (Grade 2) – Matter is Neither Created Nor Destroyed — Acids and Bases
Don’t miss our FREE chemistry packet from last year:
- Chemistry Experiments for Kids (Grade 2) – Mixtures, Chromatography, DNA Kit
- Chemistry Unit: Periodic Table – The chemistry unit we did last year as we learned about the main groups of the periodic table
- Alkali Metals — reactive
- Alkaline Earth Metals
- Halogens
- Noble Gases
- Chemistry Unit: Bohr Diagrams (this post has a free printable)
- Explosion of Colors in Milk Experiment and Other Chemistry Fun!
- Chemistry Unit: The Size of Atoms
- States of Matter: Solid, Liquid, Gas — Learning Activities
- Chemistry: Molecule Movement Experiment and Chemistry Review Worksheet These notebook pages are free.
- Science Experiments: Water Molecule Attraction
- Building Molecules Chemistry Activity This also has some free notebook pages about building molecules:

See you again soon here or Homeschool Den Facebook page. Don’t forget to Subscribe to our Homeschool Den Newsletter! ~Liesl